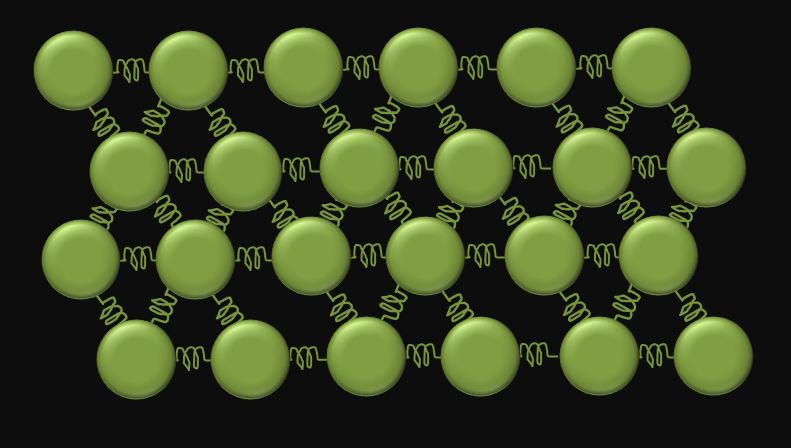
Kinetic-Molecular Theory of Solids
|
According to the Kinetic-Molecular Theory of matter, a substance in the solid phase is made up of atoms and/or molecules which are close together and which are unable to move relative to one another. In other words, the particles in a solid are bound to one another. This does not mean that the particles in a solid are not moving. The particles in a solid are moving; they just are not moving relative to one another.
The KMT of solids explains why a substance in the solid state has a definite volume and a definite shape and why solids are relatively in-compressible. A solid has a definite volume because the particles in a solid stay close together, and a solid has a definite shape because the particles in a solid cannot move around relative to one another. Solids are relatively in-compressible because the particles in a solid are close together and cannot be forced much closer together. |
|