|
Now, we must take a measurement and calculate the volume of the box holding the gas molecules.
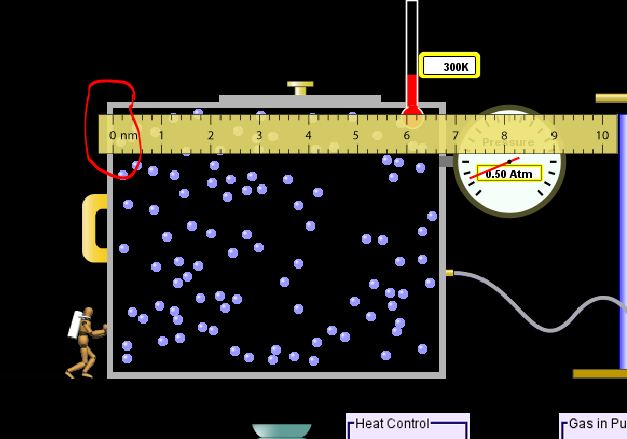
Grab the ruler in the upper left-hand corner of the simulation and drag it with your cursor until the zero marking on the ruler is lined up with the inside of the left-hand wall of the box. See the image at the right. Click on the image to enlarge it. Use the ruler to measure the width of the box from inside wall to inside wall. The width should measure about 6.62 nm. |
Now, we will calculate the volume of the box assuming its height is 6.62 nm and its depth (the dimension which goes into the page) is also 6.62 nm. The volume of a box is its (measured width) x (height) x (depth). In this case, the volume of the box is (6.62 nm) x (6.62 nm) x (6.62 nm) = 290 nm cubed. I have rounded here, but you should keep all of the digits in your calculator until after you have multiplied the volume you found with the pressure gauge reading.
Click HERE to input the volume of the box and the reading on the pressure gauge in your data table. Note: The pressure on the gauge will fluctuate; watch the pressure gauge for about 30 seconds to determine an average reading on the gauge.
Then multiply the volume by the pressure and input that into the appropriate column in the data table.
Click HERE to input the volume of the box and the reading on the pressure gauge in your data table. Note: The pressure on the gauge will fluctuate; watch the pressure gauge for about 30 seconds to determine an average reading on the gauge.
Then multiply the volume by the pressure and input that into the appropriate column in the data table.
Use your cursor to drag the left-hand wall of the box to the right until the width of the box is 6.40 nm. Calculate the new volume of the box using a width of 6.40 nm, a height of 6.62 nm, and a depth of 6.62 nm. Let the pressure gauge settle, and then determine the pressure reading at the new box volume. Input the new volume and new pressure in the data table HERE. Then, multiply the new volume by the new pressure, and input the product in the data table.
Repeat for box widths of 6.20 nm, 5.60 nm, and 5.00 nm. Don't forget to input your data and calculations into the data table HERE.
Repeat for box widths of 6.20 nm, 5.60 nm, and 5.00 nm. Don't forget to input your data and calculations into the data table HERE.
Look back at your data table and see which conclusion(s) you can draw about the pressure and volume of an ideal gas when held at a constant temperature and pressure. Unscramble the sentence below to find out if you are correct.
Pressure and Volume ConclusionSelf-Quiz
Mixed-up sentence exercise
Put the parts in order to form a sentence. When you think your answer is correct, click on "Check" to check your answer. If you get stuck, click on "Hint" to find out the next correct part.